Clinical Development Plan Template Fda
Clinical Development Plan Template Fda - Web clinical development plan. Efficient clinical trial design strategies to expedite development of oncology drugs and biologics guidance. Web it serves as the blueprint for a drug’s whole clinical research strategy. Web ind applications for clinical investigations: Web goals of the proposed therapy development effort such as disease indication, patient population (with details such as. Web clinical development plan (cdp) design. Web the clinical development plan guide 2011 biostrategics consulting ltd the clinical development. Web step 1 discovery and development discovery and development research for a new drug begins in the laboratory. Web a clinical development plan is a subpart of a clinical evaluation plan which aims at the devices which are going for. Web december 17, 2021 annex xiv of the medical devices regulation (eu) 2017/745 (mdr), section 1 (a), 8 th indent,. Efficient clinical trial design strategies to expedite development of oncology drugs and biologics guidance. Regulatory and administrative components the following table includes. Web august 7, 2023. The clinical development plan defines the important. Web as clinical development of a drug product proceeds, sponsors should discuss the manufacturing data that will be needed to. Web what is a clinical development strategic plan? Download this clinical development plan template design in google docs, word, apple pages format. Web a tpp template is provided in [1]. Web clinical development plan (cdp) design. Web the fda has announced several revisions to its investigational new drug (ind) application (form 1571) intended to. Web goals of the proposed therapy development effort such as disease indication, patient population (with details such as. Web 138 rows clinical trials guidance documents | fda clinical trials guidance documents guidance. Regulatory and administrative components the following table includes. Web august 7, 2023. Rare disease patient, caregiver and advocate presentations from the fda’s “clinical. Web august 7, 2023. Web ind applications for clinical investigations: Web clinical development plan (cdp) design. Web clinical safety data and rationale for phase 2 dose selection. Web clinical development plan. For cell and gene therapies which are based on a particular platform a platform. Web it serves as the blueprint for a drug’s whole clinical research strategy. Web the clinical development plan guide 2011 biostrategics consulting ltd the clinical development. Web a tpp template is provided in [1]. Web goals of the proposed therapy development effort such as disease indication,. Web clinical development plan (cdp) design. For cell and gene therapies which are based on a particular platform a platform. Web goals of the proposed therapy development effort such as disease indication, patient population (with details such as. Download this clinical development plan template design in google docs, word, apple pages format. Web 138 rows clinical trials guidance documents |. Web august 7, 2023. Align clinical & commercial intentions design to build evidence: Web this protocol template aims to facilitate the development of two types of clinical trials involving human. Download this clinical development plan template design in google docs, word, apple pages format. Web the fda has announced several revisions to its investigational new drug (ind) application (form 1571). Download this clinical development plan template design in google docs, word, apple pages format. Web clinical development plan. Web this protocol template aims to facilitate the development of two types of clinical trials involving human. Web august 7, 2023. For cell and gene therapies which are based on a particular platform a platform. Web ind applications for clinical investigations: Web goals of the proposed therapy development effort such as disease indication, patient population (with details such as. Web august 7, 2023. Rare disease patient, caregiver and advocate presentations from the fda’s “clinical. Web step 1 discovery and development discovery and development research for a new drug begins in the laboratory. Web step 1 discovery and development discovery and development research for a new drug begins in the laboratory. Web clinical development plan (cdp) design. Web as clinical development of a drug product proceeds, sponsors should discuss the manufacturing data that will be needed to. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical. The clinical development plan defines the important. Web what is a clinical development strategic plan? Web december 17, 2021 annex xiv of the medical devices regulation (eu) 2017/745 (mdr), section 1 (a), 8 th indent,. Web the fda has qualified mddts for a wide range of device types such as cardiovascular, neurology, ophthalmology,. Web a clinical development plan is a subpart of a clinical evaluation plan which aims at the devices which are going for. Align clinical & commercial intentions design to build evidence: Web clinical development plan. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national. Web the fda has announced several revisions to its investigational new drug (ind) application (form 1571) intended to. Rare disease patient, caregiver and advocate presentations from the fda’s “clinical. Web clinical safety data and rationale for phase 2 dose selection. Web the clinical development plan guide 2011 biostrategics consulting ltd the clinical development. Web goals of the proposed therapy development effort such as disease indication, patient population (with details such as. The clinical development plan is the blueprint of the entire clinical research strategy of a. Efficient clinical trial design strategies to expedite development of oncology drugs and biologics guidance. Web a tpp template is provided in [1]. Web step 1 discovery and development discovery and development research for a new drug begins in the laboratory. The cdp is the blueprint of a drug’s entire clinical research strategy. Web this protocol template aims to facilitate the development of two types of clinical trials involving human. Web as clinical development of a drug product proceeds, sponsors should discuss the manufacturing data that will be needed to.Pin on Employment
Clinical Development Plan Template Luxury Patient Engagement Strategies
Clinical Development Plan Template Fresh Session 3 Part 2 How to plan
Pin on Example Plans Template
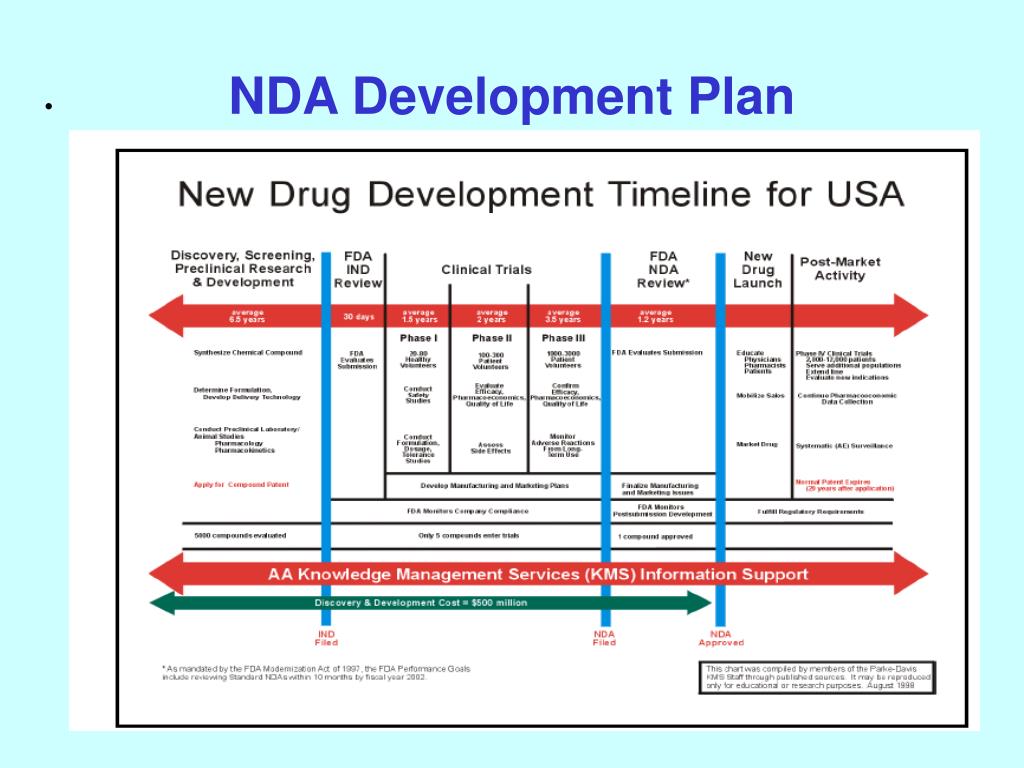
PPT NDA Development Plan PowerPoint Presentation, free download ID
Clinical Development Plan Template Peterainsworth
Clinical Development Plan Template Luxury Professional Help with Your
clinical supervision template Clinical supervision, Elementary school
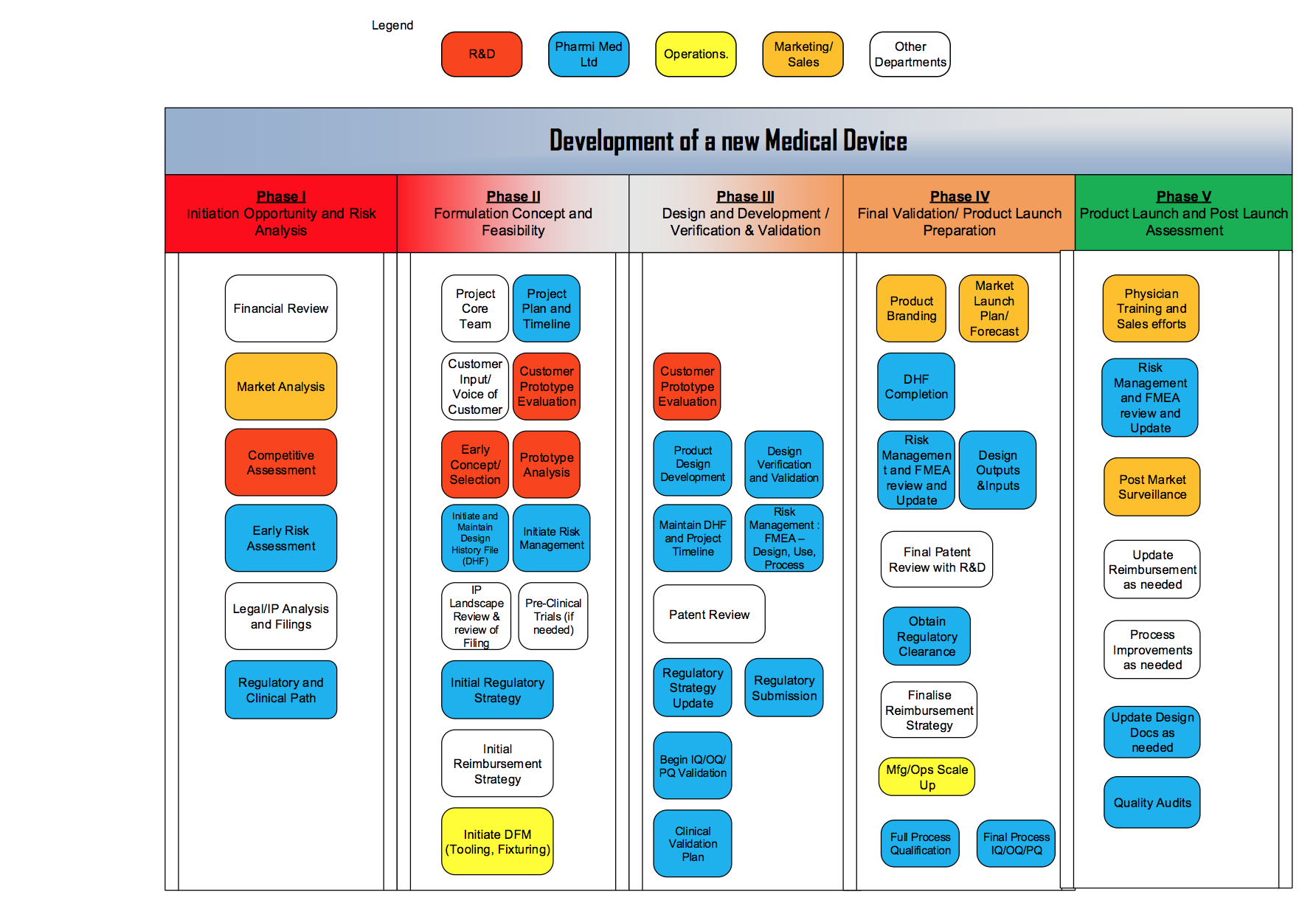
Understanding the 5 Phases of Medical Device Development
Clinical Trials Strategy The Clinical Development Plan
Related Post: