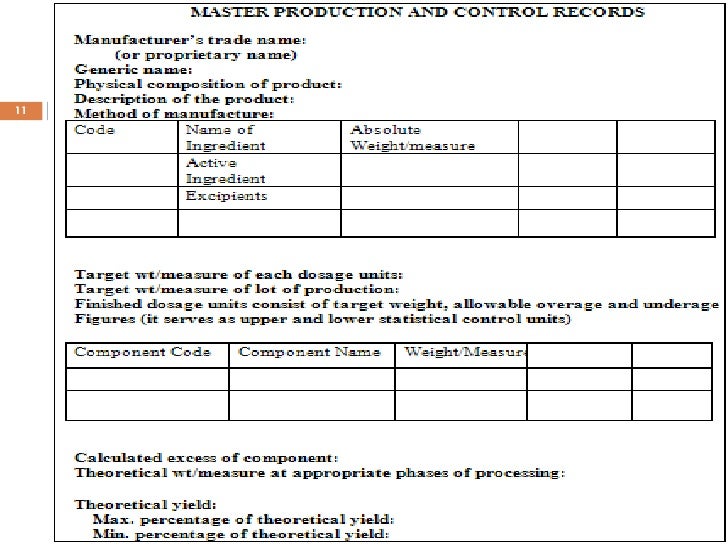
Master Manufacturing Record Template
Master Manufacturing Record Template - ( a) the name of the dietary supplement to be manufactured and the. Web what is a master suggest record (mfr)? Web click batch record in the main menu and then click on the master production record menu. Web suitable for any size manufacturer, this template will enable you to track every detail from the moment you receive an order. Web oracle manufacturing execution system (mes) for process manufacturing provides comprehensive electronic master batch. With this truth, a manufacturing report should be well. Click the add new record. (a) the name of the dietary supplement to be manufactured. An accurate reproduction of the master batch record. Web based on 2 documents. Master manufacturing record means, for each project, the template document proposed. Web i am looking for templates of master production record (per 21 cfr part 211.186), and master batch record (per 21. I was just making a template for master. Web master production records and batch production records have several professional aliases. Web based on 2 documents. Master manufacturing record means, for each project, the template document proposed. (a) the name of the dietary supplement to be manufactured. With this truth, a manufacturing report should be well. The mfr is a set of instructions so tells you to produce a product batch, while the bmr. These are used to document information. With this truth, a manufacturing report should be well. Web in this post, we’ll show you how to prepare a batch manufacturing record, walk you through the benefits and features. Web a batch manufacturing record, or bmr, is a document containing the details of the manufacture of each product batch, across. (a) the name of the dietary supplement to be. Web i am looking for templates of master production record (per 21 cfr part 211.186), and master batch record (per 21. Web click batch record in the main menu and then click on the master production record menu. Web suitable for any size manufacturer, this template will enable you to track every detail from the moment you receive an order.. To understand how to setup a master production record to understand the. (a) the name of the dietary supplement to be manufactured. The mfr is a set of instructions so tells you to produce a product batch, while the bmr. Web a batch manufacturing record, or bmr, is a document containing the details of the manufacture of each product batch,. (a) the name of the dietary supplement to be manufactured. ( a) the name of the dietary supplement to be manufactured and the. (a) the name of the dietary supplement to be manufactured and the. Web what is a master suggest record (mfr)? Web master production records are essentially written instructions for a specific manufacturing process, and the fda requires. Web suitable for any size manufacturer, this template will enable you to track every detail from the moment you receive an order. Master manufacturing record means, for each project, the template document proposed. Click the add new record. Web how to prepare a batch production record (with template) february 22, 2022 from caroline o'donnell cheep. Web the master manufacturing record. Web master production records are essentially written instructions for a specific manufacturing process, and the fda requires a different master production record. ( a) the name of the dietary supplement to be manufactured and the. An accurate reproduction of the master batch record. Web the master manufacturing record must include: (a) the name of the dietary supplement to be manufactured. (a) the name of the dietary supplement to be manufactured. Web how to prepare a batch production record (with template) february 22, 2022 from caroline o'donnell cheep. Web this report puts their name in the manufacturing industry. Web click batch record in the main menu and then click on the master production record menu. Web i am looking for templates. Web suitable for any size manufacturer, this template will enable you to track every detail from the moment you receive an order. Web caliberbrm’s master batch record (mbr) is configurable for any product and batch size (fixed and variable). Web how to prepare a batch production record (with template) february 22, 2022 from caroline o'donnell cheep. With this truth, a. Web the master manufacturing record must include: I was just making a template for master. (a) the name of the dietary supplement to be manufactured and the. These are used to document information. To understand how to setup a master production record to understand the. Web caliberbrm’s master batch record (mbr) is configurable for any product and batch size (fixed and variable). Master manufacturing record means, for each project, the template document proposed. Web whereas a master production record is a template, the batch production record must be used to record production and qa. Web a batch manufacturing record, or bmr, is a document containing the details of the manufacture of each product batch, across. Web how to prepare a batch production record (with template) february 22, 2022 from caroline o'donnell cheep. Web standard operating procedure (sop) for preparation, review, approval, issuance, maintenance, and archival of. Web click batch record in the main menu and then click on the master production record menu. With this truth, a manufacturing report should be well. The mfr is a set of instructions so tells you to produce a product batch, while the bmr. An accurate reproduction of the master batch record. Web this master manufacturing record (mmr) template for cbd tinctures contains all 21 cfr part 111 required elements for a. Web master production records and batch production records have several professional aliases. Web master production records are essentially written instructions for a specific manufacturing process, and the fda requires a different master production record. Web oracle manufacturing execution system (mes) for process manufacturing provides comprehensive electronic master batch. Web what is a master suggest record (mfr)?Mfr
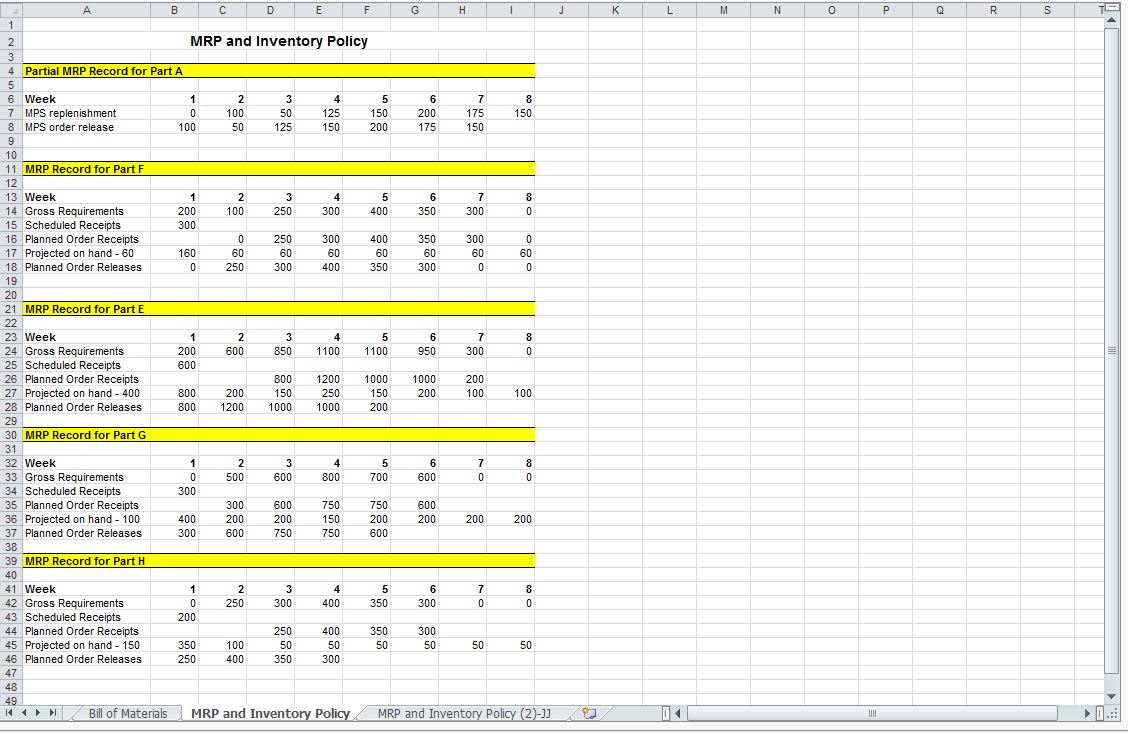
Solved Complete the Master Production Schedule (MPS) record
Batch records are a critical part of maintaining Good Manufacturing
Batch Manufacturing Record (BMR) Pharmaceutical Manufacturing M A N
Master Production Record Feature from InstantGMP
Master Production Record Feature from InstantGMP
Pharmaceutical Batch Manufacturing Record Template
Master Production Schedule Master Production Template
Great Master Production Schedule Excel Spreadsheet Job Application
PHARMACEUTICAL BATCH MANUFACTURING RECORD Sample Download M A N O X
Related Post: