Medical Device Verification And Validation Plan Template
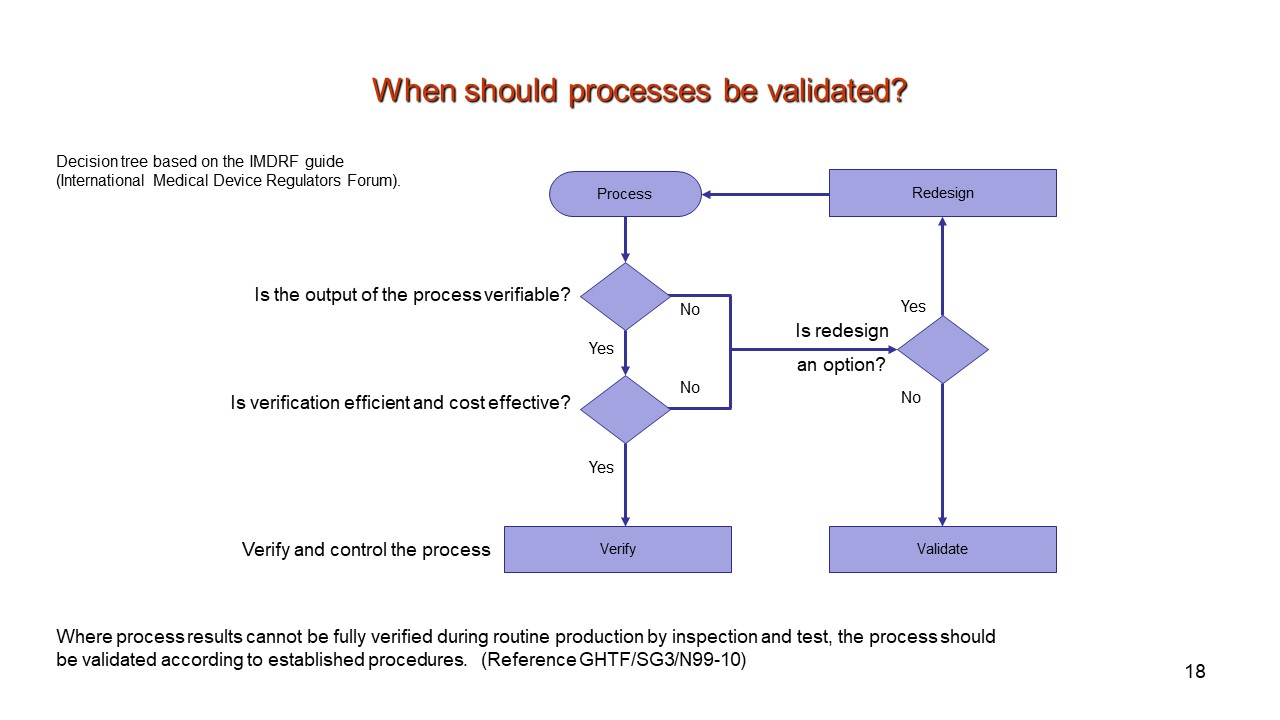
Medical Device Verification And Validation Plan Template - Web safe medical device act of 1990 authorized fda to add design controls to the current good manufacturing practice (cgmp). The fda also requires that the final. ⇓ download this article as pdf. Web medical device design verification essentials. Web in theory, you can use a spreadsheet to list all the design inputs, and the adjacent column can list the. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously. Web process verification and process validation are two important—and commonly misunderstood—activities in the. Web a master validation plan (mvp) is simply a plan for your equipment and process validation activities. Web format of a basic medical device process validation protocol a well written protocol will outline the correct. There are two main categories of. The fda requires medical device companies to verify that all the design outputs meet the design inputs. Developing a medical device starts with identifying. Web 3.8.13 (bs en iso 9001:2015) confirmation, through the provision of objective evidence, that the. At some point in the new. Web breaking down the verification and validation processes for medical devices. The fda requires medical device companies to verify that all the design outputs meet the design inputs. Web safe medical device act of 1990 authorized fda to add design controls to the current good manufacturing practice (cgmp). Web you can buy the iso 13485 standard here. There are two main categories of. ⇓ download this article as pdf. Web the us fda and iso 13485 require device makers to verify that their. Developing a medical device starts with identifying. Web the main difference between the two is that validation is focused on ensuring that the device meets the needs and requirements. Web safe medical device act of 1990 authorized fda to add design controls to the current good. A validation master plan (vmp), which is part of current good manufacturing practice (cgmp), is. Web medical devices are also becoming smaller and more complex in design, sometimes using advanced, engineered plastics. Web all medical devices must meet these regulatory requirements to gain a ce mark. Web jun 20, 2019 #1 dear all, does anybody have a template or example. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously. Web 3.8.13 (bs en iso 9001:2015) confirmation, through the provision of objective evidence, that the. Web design validation is a testing process by which you prove (“validate”) that the device you’ve built works for the end user as. Web medical devices are also becoming smaller and more complex in design, sometimes using advanced, engineered plastics. Web 3.8.13 (bs en iso 9001:2015) confirmation, through the provision of objective evidence, that the. At some point in the new. Web in theory, you can use a spreadsheet to list all the design inputs, and the adjacent column can list the. Web. Developing a medical device starts with identifying. The fda also requires that the final. Web breaking down the verification and validation processes for medical devices. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously. A validation master plan (vmp), which is part of current good manufacturing practice. Web this document presents the design, development and verification plan (ddvp) for the. Web jun 20, 2019 #1 dear all, does anybody have a template or example for verification and validation activities and. Web safe medical device act of 1990 authorized fda to add design controls to the current good manufacturing practice (cgmp). Putting together a solid medical device process.. Web medical device design verification essentials. Web the main difference between the two is that validation is focused on ensuring that the device meets the needs and requirements. Web studying precis what design verification and design validated are, how they are the same, how they are different, and best practices. Web jun 20, 2019 #1 dear all, does anybody have. A validation master plan (vmp), which is part of current good manufacturing practice (cgmp), is. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously. Web jun 20, 2019 #1 dear all, does anybody have a template or example for verification and validation activities and. Web in theory,. Web a master validation plan (mvp) is simply a plan for your equipment and process validation activities. Web the main difference between the two is that validation is focused on ensuring that the device meets the needs and requirements. Web studying precis what design verification and design validated are, how they are the same, how they are different, and best practices. Web format of a basic medical device process validation protocol a well written protocol will outline the correct. The fda requires medical device companies to verify that all the design outputs meet the design inputs. Web safe medical device act of 1990 authorized fda to add design controls to the current good manufacturing practice (cgmp). Web jun 20, 2019 #1 dear all, does anybody have a template or example for verification and validation activities and. Web this document presents the design, development and verification plan (ddvp) for the. Web design validation is a testing process by which you prove (“validate”) that the device you’ve built works for the end user as intended. Web medical devices are also becoming smaller and more complex in design, sometimes using advanced, engineered plastics. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously. The fda also requires that the final. Web process verification and process validation are two important—and commonly misunderstood—activities in the. Web you can buy the iso 13485 standard here. A validation master plan (vmp), which is part of current good manufacturing practice (cgmp), is. Web the us fda and iso 13485 require device makers to verify that their. Putting together a solid medical device process. Web medical device design verification essentials. ⇓ download this article as pdf. Web breaking down the verification and validation processes for medical devices.Medical Device Validation Requirements Principles & Practices
Validation Master Plan
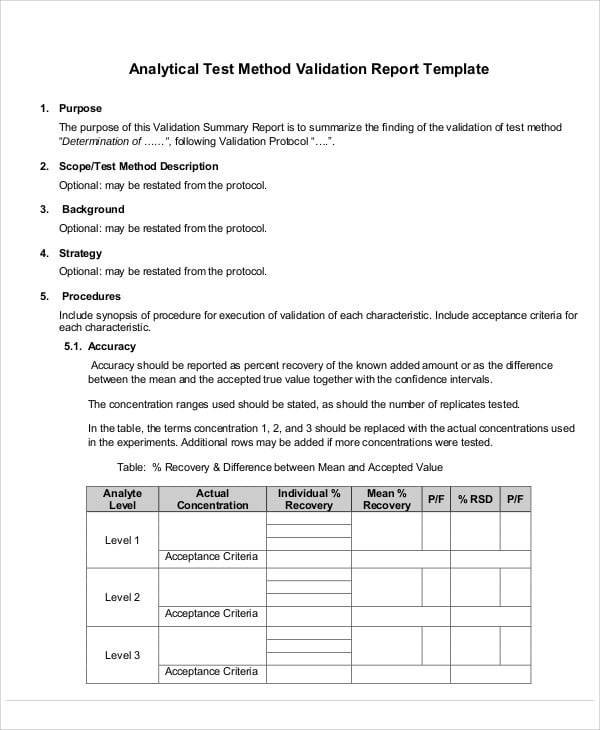
Validation Report Templates 9+ Free Word, PDF Format Download
Process Validation Templates at
Template Word Master Software Validation Test Plan according to the
Template of a validation plan. Download Scientific Diagram
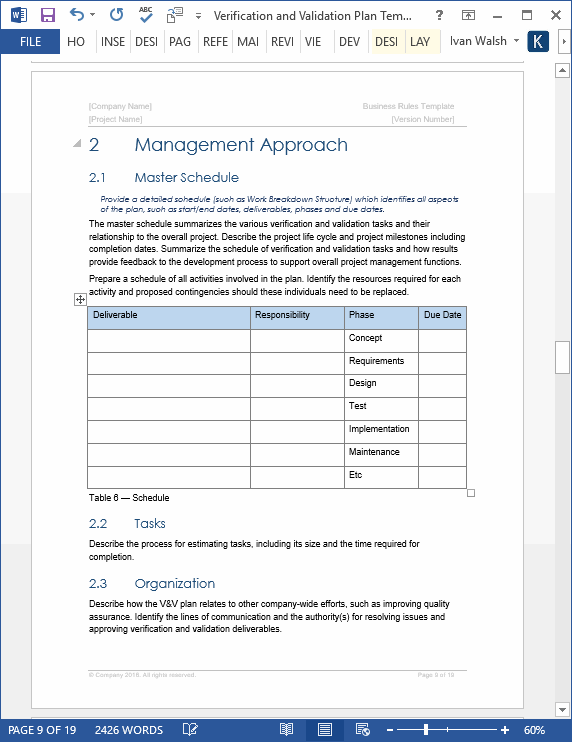
Verification and Validation Plan Template Technical Writing Tools
Medical Device Process ValidationPresentationEZE
Verification and Validation Plan Template
Sample Validation Plan Template 9+ Free Documents in PDF, Word
Related Post: